Traditional clinical trials take place at designated study sites. Investigators and their personnel usually perform the procedures regarding the study. Patients within the clinical trial need to visit the site to meet with them, causing struggles for the patients. While technology in every life aspect advances, so does healthcare technology. Several of these technological developments are meant to solve nowadays challenges in the clinical trial sector. That’s why the 21st Century Cures Act was signed into US law on December 13, 2016, to implement the new technology into laws and regulations. It authorized 500 $ million over nine years to help the U.S. Food and Drug Administration (FDA) cover the costs. However, what exactly will be new, what is already there, and what is going to change in the future?
Decentralized clinical trials (DCT) are already the go-to option for clinical. From a historical perspective focused on regulations, they have come a long way. It started in 2007 with an official FDA guidance document, which provides recommendations regarding the use of Computerized Systems Used in Clinical Investigations. This document is the basis for modern technology within a clinical study.
Mobile devices and biosensors grow in importance
The next step took place six years later. To modernize clinical investigations, the guidance for Electronic Source Data in Clinical Investigations promotes capturing source data in electronic form. It’s meant to assist in making sure of the reliability, quality, integrity, and traceability of data from the source to electronic regulatory submission.
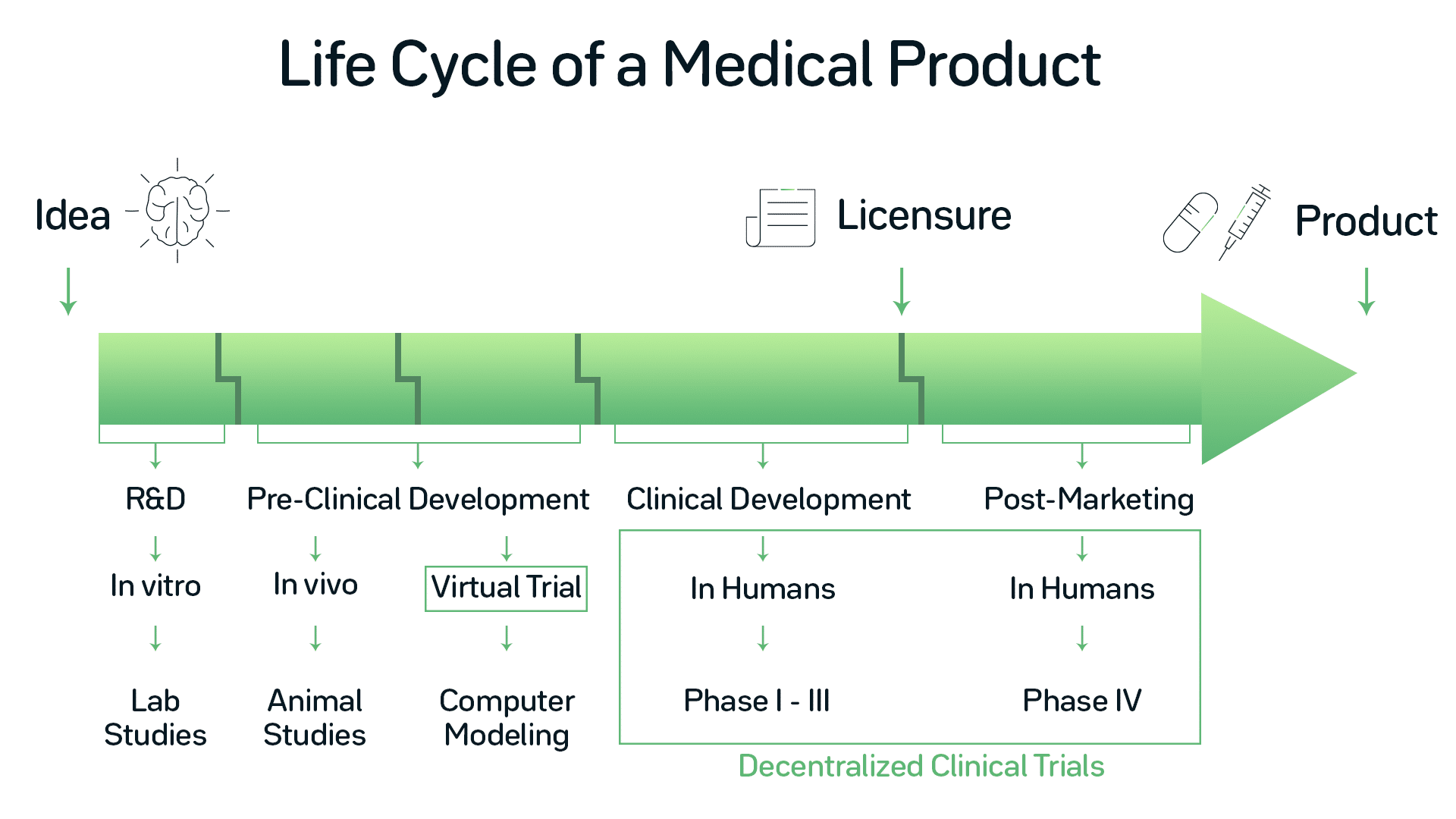
The latest development is the FDA guidance document to the Use of E-records and E-signatures in Clinical Investigations Under 21 CFR Part 11. The FDA published it as a draft in 2017. The paper outlines the scope and application of part 11 requirements for clinical investigations of medical products. Besides, it covers the topic of electronic data transmission and audit trails from electronic devices. Also, some efforts were made in regards to digital health technologies. These include the use, for example, of mobile devices and biosensors in clinical trials.
Next: Decentralized Clinical Trials
There is a considerable advance in electronic communication tools, data transmission, digital technology, and development in the field of biosensors. These developments open new possibilities for information exchange between investigators and researchers. Locations remote from the investigator’s home become available as a convenient option for home-based clinical trials.
Also, the spectrum of clinical trials becomes wider. The classical Controlled Clinical Trial (CCT) is no longer the only option. Besides, the DCT makes it possible to target a whole new study subject group. People who can’t leave their homes or can’t regularly travel to far away study sites get the chance to be part of the medical research. This approach will not only improve the subjects’ likelihood of finding a cure but also lead to more reliable, diverse and valid study results. As a third option, Hybrid Models are a middle way of CCT and DCT.
Trials of the future will integrate the study subject’s existing healthcare system. The integration maximizes convenience for study subjects. Also, it takes advantage of already existing services and data sources.
A relatively new part of DCT is the so-called virtual clinical trials. In this case, the whole study takes place without any physical visit of an investigator. After a short onboarding phase, in which a “site concierge” installs and explains all needed technology to the study subject, there is no need for further physical contact with the study staff. Of course, this varies from study to study and can not be applied to every trial.
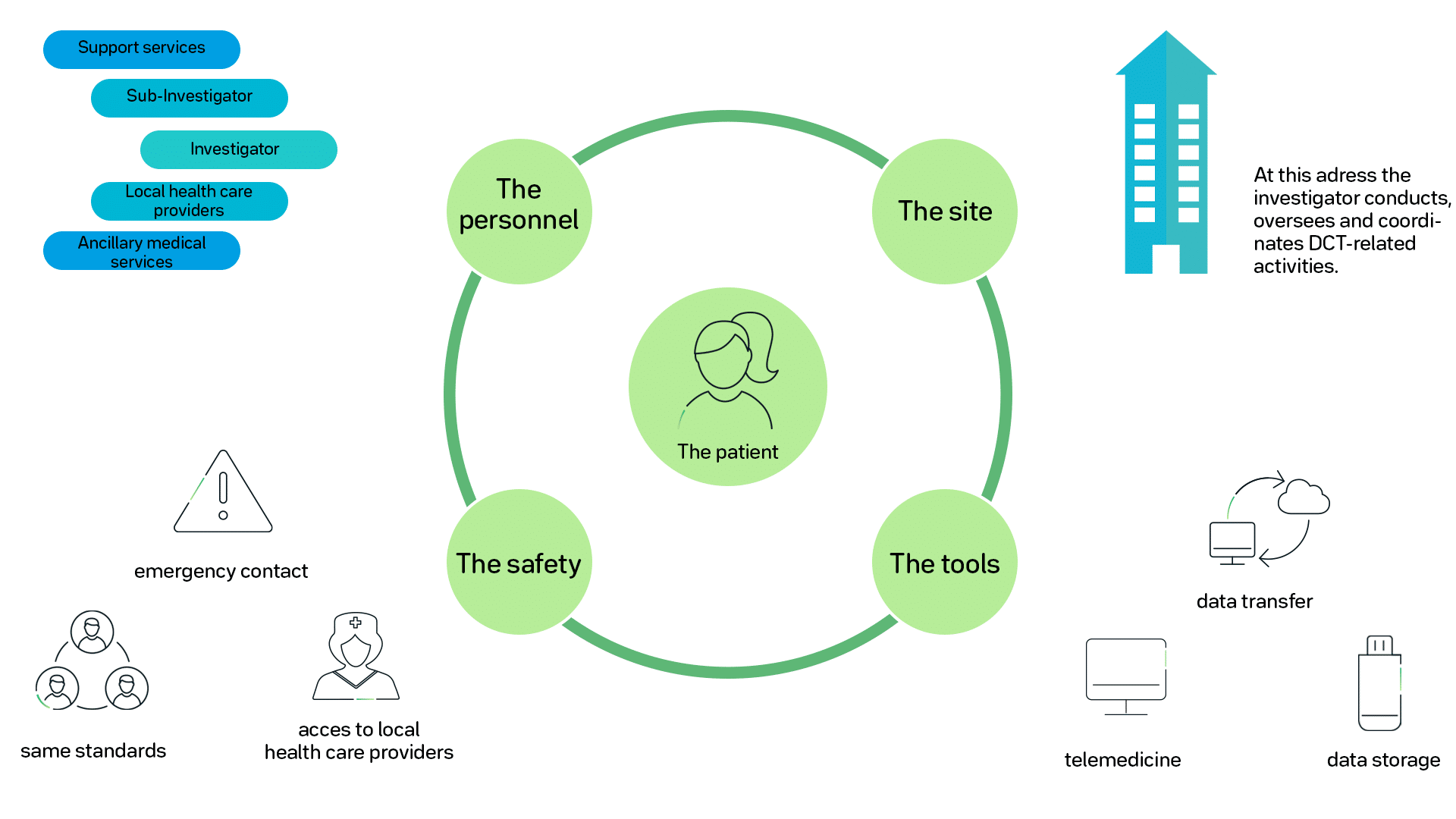
Tools and regulations
The study subjects will use videoconferences with their investigator, have internet-based tools for reporting, and mobile technology tools, including biosensors. This network of information gathers a ton of data, which is immediately visible for the responsible investigators and researchers. At the same time, local healthcare providers, such as pharmacies, clinics, and home-based healthcare services, are also included in collecting data about the study subject.

“The agency has worked closely with stakeholders, including the Clinical Trial Transformation Initiative, to identify innovative trial designs, evaluate the role of decentralized clinical trials and mobile technologies, and help validate novel endpoints that can enable trials to generate reliable evidence needed to assess product safety and efficacy more efficiently.”
The pharmacy, for example, will get an alert if the subject is close to running out of medicine. Reports from homecare are part of the data collection, giving more profound insights into the subjects’ well-being. With the help of an intelligent refrigerator for medical supplies, adherence and temperature control can always be monitored.
The regulations that apply to DCT or hybrid models of clinical trials will be the same that already use for any clinical trial. Data quality and integrity must fulfill all federal regulation standards. The safety and confidentiality of the study subject are also bound to the same regulations.
